 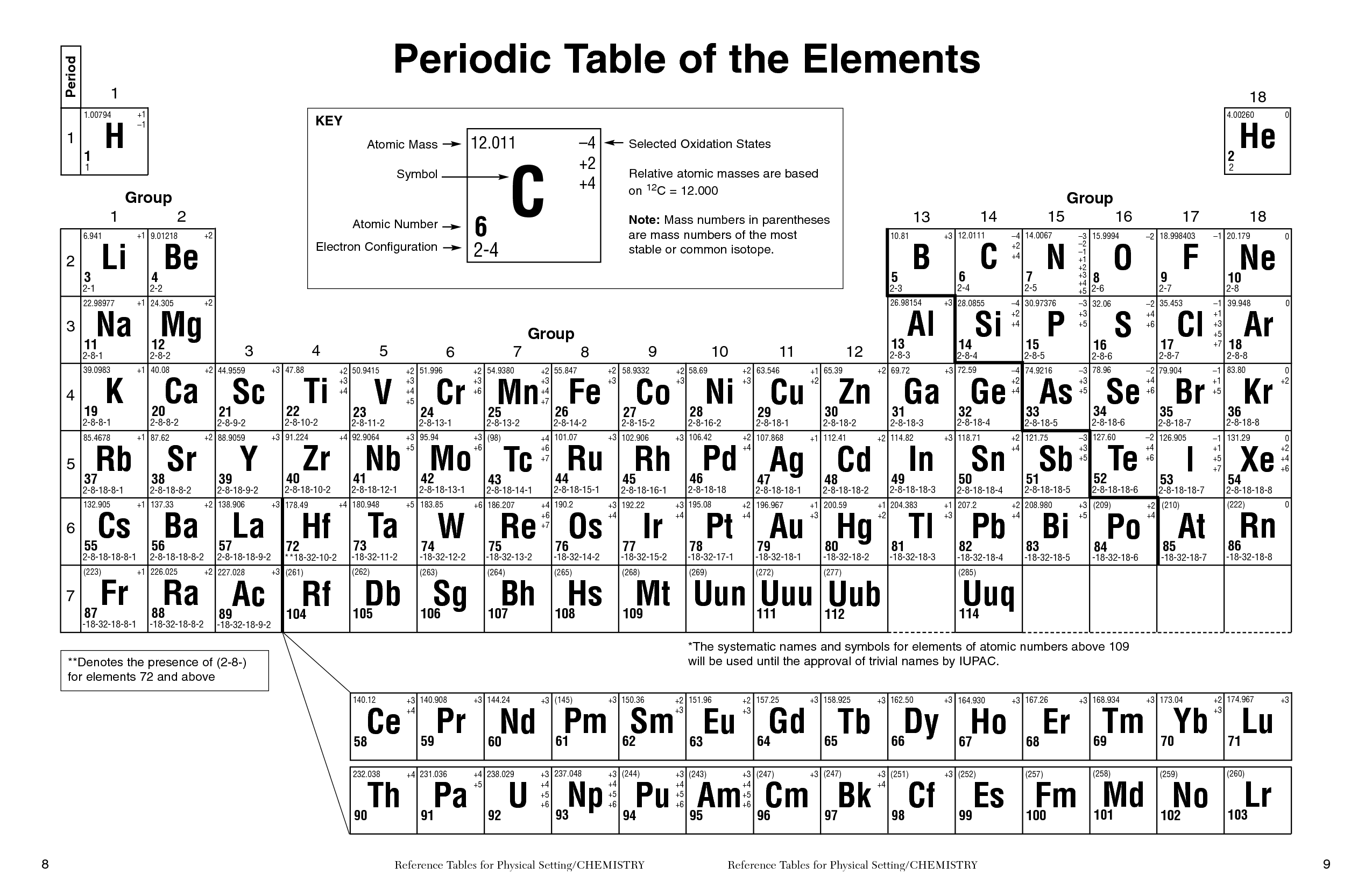
Similarly, as we proceed across the row, the increasing nuclear charge is not effectively neutralized by the electrons being added to the 2 s and 2 p orbitals. In chemistry, periodicity refers to repeating trends in element properties on the periodic table. Consequently, beryllium is significantly smaller than lithium. This means that the effective nuclear charge experienced by the 2 s electrons in beryllium is between +1 and +2 (the calculated value is +1.66). It is a physical science within the natural sciences that studies the chemical elements that make up matter and compounds made of atoms, molecules and ions: their composition, structure, properties, behavior and the changes they undergo during reactions with other substances. (More detailed calculations give a value of Z eff = +1.26 for Li.) In contrast, the two 2 s electrons in beryllium do not shield each other very well, although the filled 1 s 2 shell effectively neutralizes two of the four positive charges in the nucleus. Chemistry is the scientific study of the properties and behavior of matter. Thus the single 2 s electron in lithium experiences an effective nuclear charge of approximately +1 because the electrons in the filled 1 s 2 shell effectively neutralize two of the three positive charges in the nucleus. Although electrons are being added to the 2 s and 2 p orbitals, electrons in the same principal shell are not very effective at shielding one another from the nuclear charge. The nonmetals include the group called the nonmetals, as well as the halogens and the noble gases. These elements are located on the righthand side of the table, with the exception of hydrogen. All have a filled 1 s 2 inner shell, but as we go from left to right across the row, the nuclear charge increases from +3 to +10. The nonmetals are a small group of elements on the periodic table. The atoms in the second row of the periodic table (Li through Ne) illustrate the effect of electron shielding. All 118 elements of the Modern Periodic Tabe is listed below. Mendeleev’s Periodic Table was published in the German Journal of chemistry in 1869. The greater the effective nuclear charge, the more strongly the outermost electrons are attracted to the nucleus and the smaller the atomic radius.Ītomic radii decrease from left to right across a row and increase from top to bottom down a column. During his time only around half of the elements known to us now were known, and most of the information known about the elements were inaccurate. For all elements except H, the effective nuclear charge is always less than the actual nuclear charge because of shielding effects. Because of differing definitions, chemistry teachers probably ought to stay away from questions about elements/compounds and simply focus on the 118 elements of the periodic table as examples of elements. \( \newcommand\)) experienced by electrons in the outermost orbitals of the elements. By this definition, O 2 would be an element, a molecule, and a compound. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
